RADIESSE® (+)
 RADIESSE® (+) is manufactured by MERZ AESTHETICS™ and is an opaque dermal filler that contains calcium-based microspheres suspended in a water-based gel carrier that comes in a 1.5 ml syringe premixed with 0.3% powdered lidocaine. It contains no animal proteins and does not require allergy pre-testing. This product works in two ways. The gel carrier gives an immediate correction. The calcium-based microspheres act as a scaffold, stimulating your body to produce its own natural collagen. Over time, the gel carrier is absorbed and the calcium-based microspheres are metabolized by your body, leaving behind the newly created collagen. Unlike hyaluronic acid dermal fillers, RADIESSE® (+) cannot be dissolved with an enzyme.
RADIESSE® (+) is manufactured by MERZ AESTHETICS™ and is an opaque dermal filler that contains calcium-based microspheres suspended in a water-based gel carrier that comes in a 1.5 ml syringe premixed with 0.3% powdered lidocaine. It contains no animal proteins and does not require allergy pre-testing. This product works in two ways. The gel carrier gives an immediate correction. The calcium-based microspheres act as a scaffold, stimulating your body to produce its own natural collagen. Over time, the gel carrier is absorbed and the calcium-based microspheres are metabolized by your body, leaving behind the newly created collagen. Unlike hyaluronic acid dermal fillers, RADIESSE® (+) cannot be dissolved with an enzyme.
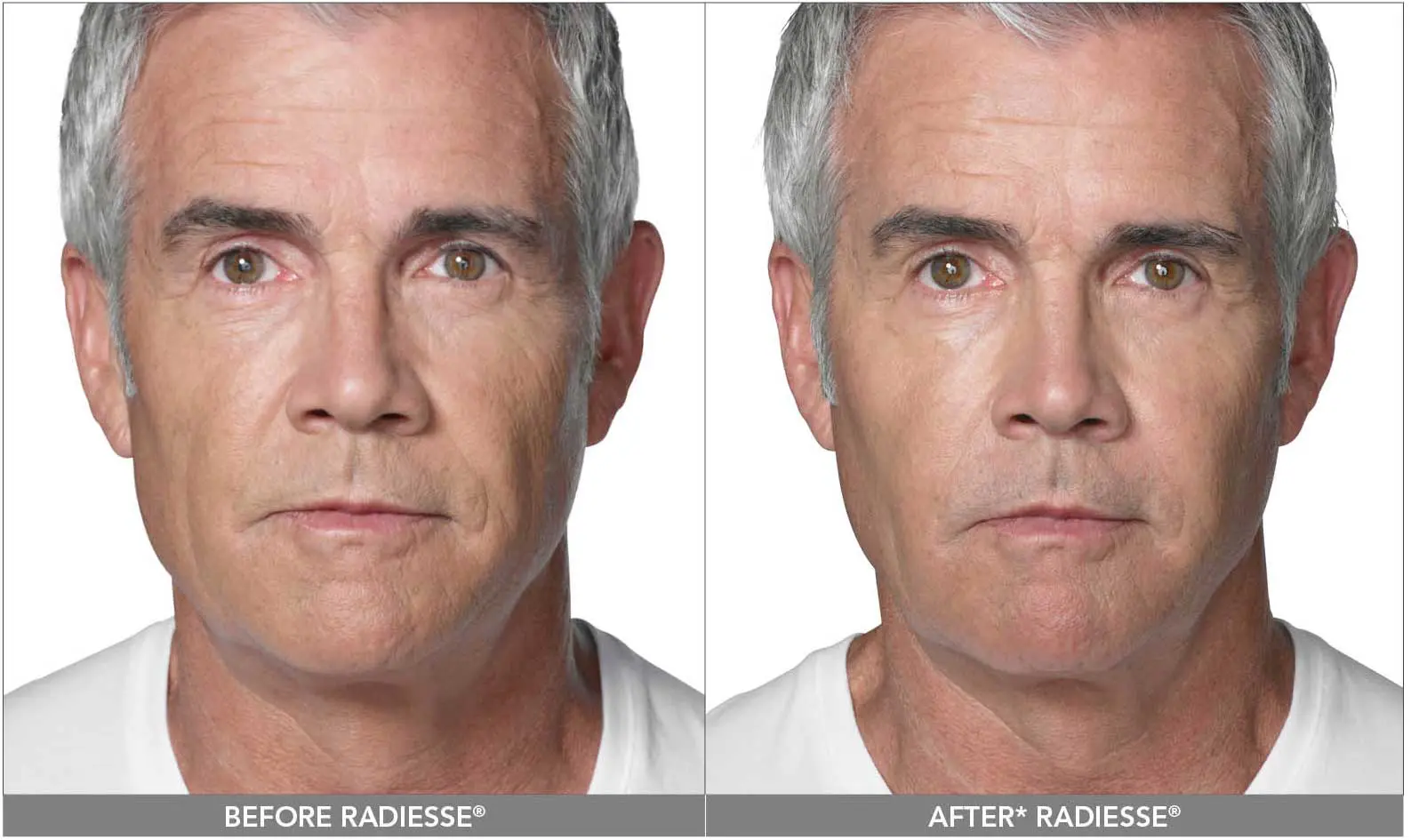 RADIESSE® (+) has the same precautions as other dermal fillers with three exceptions. The calcium-based microspheres are visible on CT scans and may be visible in standard plain radiography. It is advised to tell your treating physician or radiologist that you have had RADIESSE® (+) injected in your face before any radiological studies are performed. The second precaution is that RADIESSE® (+) has been known to activate herpes simplex and shingles in patients. Appropriate treatment should be started if any symptoms arise after an injection procedure. The third precaution is that RADIESSE® (+) should not be injected around the eyes or in the lips as the formation of nodules may occur in these areas.
RADIESSE® (+) has the same precautions as other dermal fillers with three exceptions. The calcium-based microspheres are visible on CT scans and may be visible in standard plain radiography. It is advised to tell your treating physician or radiologist that you have had RADIESSE® (+) injected in your face before any radiological studies are performed. The second precaution is that RADIESSE® (+) has been known to activate herpes simplex and shingles in patients. Appropriate treatment should be started if any symptoms arise after an injection procedure. The third precaution is that RADIESSE® (+) should not be injected around the eyes or in the lips as the formation of nodules may occur in these areas.
 RADIESSE® (+) can be used in multiple areas on the face. It can be injected deeply to lift and augment the cheeks, temples, chin and pre-jowl area. It can be injected more superficially for nasolabial folds, marionette lines and chin creases. A treatment with RADIESSE® (+) may result in a correction lasting a year or longer.
RADIESSE® (+) can be used in multiple areas on the face. It can be injected deeply to lift and augment the cheeks, temples, chin and pre-jowl area. It can be injected more superficially for nasolabial folds, marionette lines and chin creases. A treatment with RADIESSE® (+) may result in a correction lasting a year or longer.
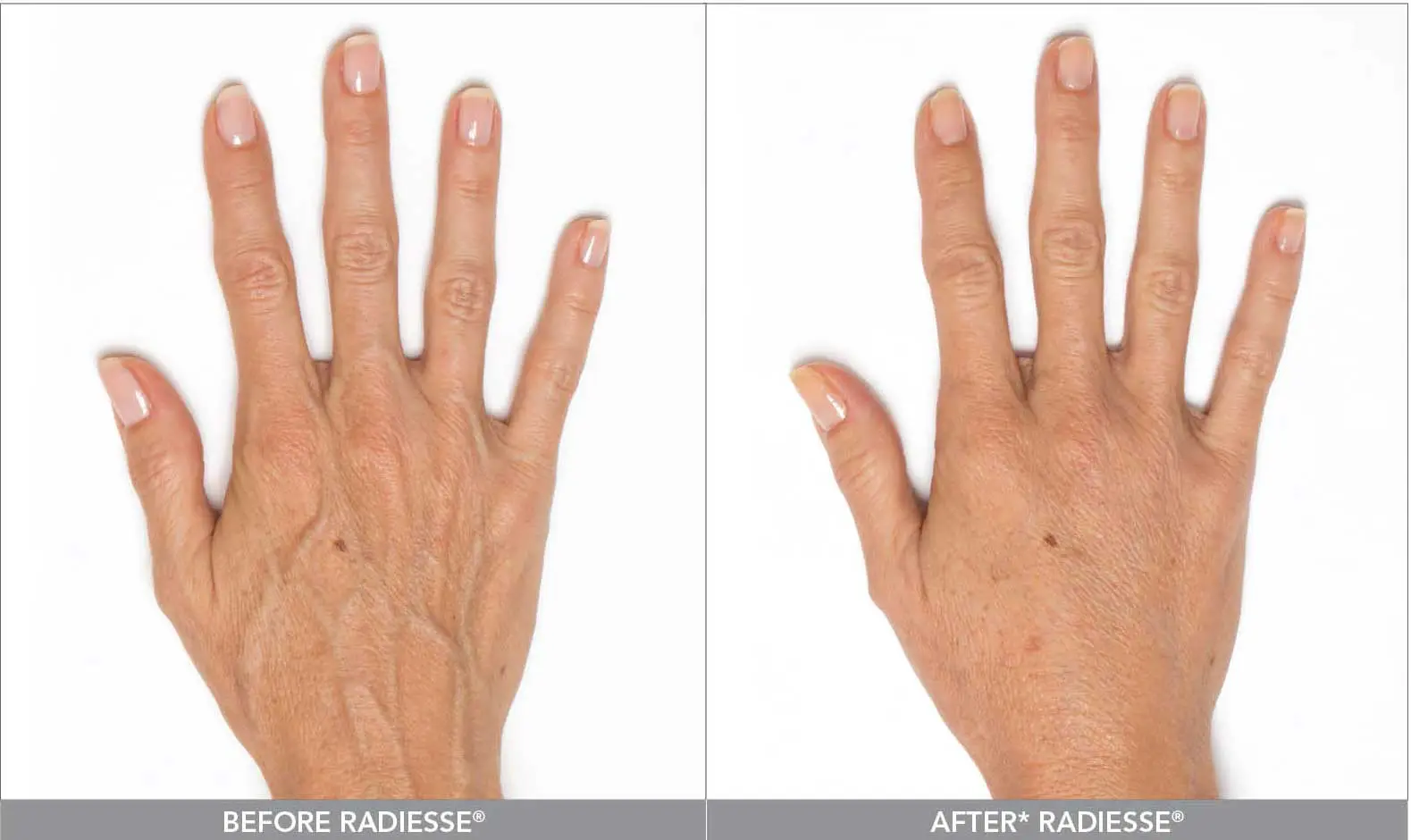 RADIESSE® (+) received FDA approval for Hand Augmentation to correct volume loss on the dorsum (back) of the hands. It is injected under the skin of the dorsal surface of the hands to lessen the appearance of visible veins and tendons due to volume loss. The result is a more youthful appearance to the back of your hands.
RADIESSE® (+) received FDA approval for Hand Augmentation to correct volume loss on the dorsum (back) of the hands. It is injected under the skin of the dorsal surface of the hands to lessen the appearance of visible veins and tendons due to volume loss. The result is a more youthful appearance to the back of your hands.
 In 2006, RADIESSE® was approved by the FDA for the treatment of Facial Lipoatrophy associated with HIV. MERZ AESTHETICS™ has a Patient Assistance Program for patients with Facial Lipoatrophy associated with HIV. This program is available for HIV patients on a limited income without insurance that would cover a restorative procedure. From our office, a simple form for this program can be filled out and submitted with proof of income. Once accepted into the program, a patient will be able to purchase up to six 1.5 ml syringes of RADIESSE® per calendar year. The syringe cost varies depending on a patient’s income. A patient is requested to cover the cost of the syringes prior to ordering, and there is an injection fee per syringe charged at the time of a treatment. The syringes do not have to be ordered all at once but can be ordered several at a time during the course of the calendar year up to the six allotted. Usually, a patient will use two to four syringes on an initial treatment to restore and correct volume loss in the face. The remaining syringes can be used over the course of the year to maintain the correction.
In 2006, RADIESSE® was approved by the FDA for the treatment of Facial Lipoatrophy associated with HIV. MERZ AESTHETICS™ has a Patient Assistance Program for patients with Facial Lipoatrophy associated with HIV. This program is available for HIV patients on a limited income without insurance that would cover a restorative procedure. From our office, a simple form for this program can be filled out and submitted with proof of income. Once accepted into the program, a patient will be able to purchase up to six 1.5 ml syringes of RADIESSE® per calendar year. The syringe cost varies depending on a patient’s income. A patient is requested to cover the cost of the syringes prior to ordering, and there is an injection fee per syringe charged at the time of a treatment. The syringes do not have to be ordered all at once but can be ordered several at a time during the course of the calendar year up to the six allotted. Usually, a patient will use two to four syringes on an initial treatment to restore and correct volume loss in the face. The remaining syringes can be used over the course of the year to maintain the correction.
Schedule a Complimentary Consultation with Willo MediSpa in Phoenix to discuss RADIESSE® (+) by calling 602-296-4477 or by contacting us below.
Love This Place!
Cathy B. – Phoenix
“Great service. I love their products!”
Long Time Client!
Bridget P. – Phoenix
“I wouldn’t go any where else then Willo MediSpa!”



 SkinMedica® Store
SkinMedica® Store